 Effect Of Growth Stage
And Variety On Spectral Radiance In Winter Wheat
Effect Of Growth Stage
And Variety On Spectral Radiance In Winter WheatEffect of Growth Stage and
Variety on
Spectral Radiance in Winter Wheat
H. Sembiring, H.L. Lees, W.R.
Raun, J.B. Solie,
M.L. Stone, G.V. Johnson, E.V. Lukina, D.A.
Cossey, J.M. LaRuffa,
C.W. Woolfold,
S.B. Phillips, and W.E. Thomason
ABSTRACT
Before sensor-based VRT (variable rate technology) can be used to reduce nitrogen input costs in winter wheat (Triticum aestivum, L.) production, several issues involved with obtaining and understanding the spectral radiance readings must be resolved. One prominent issue is the impact of crop growth stage on spectral radiance readings, and the ensuing problem of relating databases gathered at different locations and different stages of growth. In order to evaluate the impact of growth stage on spectral radiance, readings were taken from a winter wheat variety trial and two long-term nitrogen (N) and phosphorus (P) fertility trials. Spectral radiance readings were also taken at different stages of growth (Feekes 4 to Feekes 8). Results indicated a relationship exists between the normalized difference vegetative index (NDVI=(NIR-red)/(NIR+red)) and total N uptake. Small differences in five common wheat varieties over growth stages from NDVI readings were observed. Slopes from linear regression of total N uptake on NDVI were different at different stages of growth, which suggests the need for growth stage specific calibration.
INTRODUCTION
|
|
Field element size is the area on which a single rate of nitrogen or other nutrient is applied; the fundamental field element is that area which provides the most precise measure of the available nutrient where the level of that nutrient changes with distance (Solie et al., 1996). Work by Solie et al. (1996) demonstrated that the field element size where real differences are likely to exist is less than 1.96 m2. Hence, sensor-based VRT (sVRT) may be the only cost effective answer for addressing nutritional needs. In sVRT, an integrated sensor and signal processing system is used to obtain spectral radiance readings. Sensor- based VRT holds perhaps more promise for the reduction of fertilizer costs than map-based VRT due to the cost of implementing the latter (Sawyer, 1994). While improving fertilizer management via sensors has advanced, many uncharacterized variables affect spectral radiance readings. One of these variables is growth stage. By describing this variables impact, we can more effectively utilize spectral readings for the precise application of need based fertilizer rates.
Growth stage of the crop is one of the many factors affecting spectral radiance in wheat (Triticum durum Desf. var Produra) (Jackson et al.,1983), grasses (Hagger et al., 1984), and barley (Horedum vulgare, L.) (Kleman and Fagerlund, 1987). Research in barley found that the relationship between fresh biomass and IR/red changed with time, indicating the ratio was dependent on growth stage (Kleman and Fagerlund, 1987). Other research has documented why growth stage affects reflectance readings. Gausman et al. (1971) explained why reflectance changed due to growth stage in cotton. Young, compact leaves had less air spaces within the mesophyll than did older leaves, and thus, showed decreased NIR spectral radiance. Furthermore, leaf aging had a variable effect on visible light reflectance. Chlorophyll molecules were sensitive to disease and other physiological stresses (Knipling, 1970). Initially, these stresses were exhibited by changes in the visible spectral region, but not in the infrared (Knipling, 1970). Wanjura and Hatfield (1987) found that overall, a ratio was more sensitive to high levels of biomass and leaf area index (LAI), but when crops were small, normalized difference and greenness were better indicators of LAI and groundcover. Therefore, the reliability of spectral vegetative indices is dependent upon the growth stage of the crop. The objective of this paper was to evaluate the impact of growth stage on NDVI readings of winter wheat in Oklahoma.
MATERIALS AND METHODS
Three winter wheat trials were selected in the fall of 1995: a long-term N and P fertility trial at Stillwater, OK on a Kirkland soil loam (fine, mixed, thermic Udertic Paleustoll), another long-term N and P fertility trial at Lahoma, OK on a Grant silt loam (fine-mixed thermic Typic Paleustoll) and a variety by N rate trial at Hennessey, OK on a Shellabarger sandy loam (fine, loamy, mixed, thermic Udic Argiustoll). Winter wheat was planted in October and November of 1995 at 62.5 kg ha-1 (Hennessey) and 67 kg ha-1 (Stillwater and Lahoma). The variety by N rate trial consisted of a full factorial arrangement of treatments including 5 varieties and 3 preplant N rates, evaluated in a randomized complete block design. The varieties were: Tonkawa, Jagger, Karl 92, Agri-Pro Hickok, and AGSECO 7853+. Preplant N rates evaluated were 0, 84, and 168 kg N ha-1. Each plot was 15.24 m x 2.44 m. The long-term N and P fertility trial at Stillwater evaluated N rates of 0 to 135 kg ha-1 in a completely randomized block design. Also, the long-term N and P fertility trial at Lahoma evaluated N rates of 0 to 112 kg ha-1 in a completely randomized block design. Main plot size in the long-term trial at Stillwater was 18.29 m x 6.10 m and 18.29 m x 4.88 m in the long-term trial at Lahoma. Row spacing for the long-term trials was 19.05 cm at Stillwater, and 21.59 cm at Lahoma. The wheat variety Tonkawa was planted at both sites. At the variety by N rate trial at Hennessey, a 20.32 cm row spacing was used.
The long-term trial at Stillwater was sampled at Feekes growth stage 5 (sheaths of leaves strongly erected) and 7 (second node of stem formed, next to last leaf just visible). The long-term trial at Lahoma was sampled at Feekes growth stage 7 and 8 (last leaf visible, but still rolled up, ear beginning to swell), and the variety by N rate trial at Hennessey was sampled at growth stage 4 (leaf sheaths beginning to lengthen), 5, and 7. Spectral radiance readings were collected just prior to obtaining forage clippings. Photodiode detectors and interference filters for red (671 +/- 6 nm), green (550 +/- 6 nm), and NIR (780 +/- 6 nm) spectral bands with a 0.305m width by 0.075 m length spatial resolution were used. Readings were taken with a sensor mounted on the front of a John Deere 318 garden tractor. Details on sensor design can be found in Stone et al. (1996). Sensor height was adjusted according to row spacing. Sensor readings were taken from subplot areas of (variety by N rate trial at Hennessey) 0.91m x 0.61m, (long-term trial at Stillwater) 0.91m x 0.57m, and (long-term trial at Lahoma) 0.91m x 0.65m. Forage yield was determined by handclipping the entire subplot area (0.91m x 3 rows) 2 cm above the ground. The plant material was oven dried for 3 days at 700°C in a forced air oven. The amount of fresh material and forage moisture was also calculated. Total N in plant tissue was determined using a Carlo-Erba (Milan, Italy) NA 1500 dry combustion analyzer. Total N uptake was found by multiplying total N and dry biomass. NDVI was calculated for the spectral radiance readings using the following formula: (NIR - red)/(NIR + red). Analysis of variance and correlation of wet and dry biomass and total N uptake with green, red and NIR uncalibrated voltage readings, and NDVI vegetative indices were also evaluated for each trial. Initial surface (0-15cm) soil test characteristics at each site are reported in Table 1.
Results
Analysis of variance on spectral radiance, NDVI and total N uptake at different growth stages, is presented in Tables 2, 3, and 4. Results are presented by location.
Long-term N and P fertility trial, Stillwater
For all dependent variables analyzed, no significant N rate by growth stage interactions were detected, thus allowing independent interpretation of main effects (Table 2). A significant linear response to applied N was observed for N uptake and NDVI. Also, N uptake and NDVI were different with changing growth stages, which was observed for red, green, and NIR.
Long-term N & P fertility trial, Lahoma
The N rate by growth stage interaction was significant for N uptake, thus restricting main effect interpretation of this variable. However, this was not observed for NDVI or the red, green, and NIR spectral radiance readings at Lahoma (Feekes growth stages 7 and 8). A significant linear response to N rate was observed for NDVI, red, green, and NIR spectral radiance readings. Growth stage was significant for red, green and NIR spectral radiance readings. NDVI, however, was not significant for growth stage.
In the long-term trial at Lahoma, red spectral radiance and NDVI decreased and NIR increased, as N rate increased. One would expect higher N rates to produce more biomass; therefore, decreasing red and increasing NIR.
Variety by N rate trial, Hennessey
The variety by growth stage interaction was significant for N uptake (Feekes growth stages 4, 5, and 7) (Table 4). The N rate by variety interaction was significant for the red and green spectral radiances. NDVI, red, green, and NIR were significant for growth stage. No differences in NDVI or NIR were observed when evaluated over varieties or N rates.
Discussion
The variety by growth stage interaction for N uptake at Hennessey was explained by finding significant differences in variety at growth stage 4, but none at growth stage 5 (Table 4). Different varieties break dormancy at different times, and metabolism is limited when they are dormant. By growth stage 5 there was not a significant difference in N uptake values by variety, possibly because all varieties had broken dormancy by this time (February 26).
The effect of N fertilizer rates on spectral radiance were evaluated for the three sites. N rate was linear for NDVI and total N uptake in the long-term N and P fertility trial at Stilllwater and N rate was linear for NDVI in the long-term N and P fertility trial at Lahoma; however, N rate was not linear for any of the parameters in the variety by N rate trial. This was possibly due to high soil test nitrate (21.2 mg kg-1) and ammonium (18.4 mg kg-1) at this site.
NDVI was significant for growth stage in the long-term N and P fertility trial at Stillwater (growth stage 5 and 7) and the variety by N rate trial (growth stage 4, 5, and 7), but not in the long-term N & P fertility trial at Lahoma (growth stage 7 and 8). This was possibly due to the level of crop maturity at the time spectral radiance readings were taken, and proximity of the harvest dates. There was no difference in NDVI when evaluated over varieties.
A significant N rate by variety interaction was found for the red and green spectral readings. Thus, interaction was the result of having lower radiance readings for Jagger at the 84 kg N ha-1 rate when compared to all other varieties. Although this effect was significant, we cannot explain why.
Regression analysis of total N uptake on NDVI from the long-term N & P fertility trials at Stillwater and Lahoma, and the variety by N rate trial at Hennessey is presented in Figures 1, 2, and 3, respectively. Hinzman et al. (1986) presented a similar curve in their work for leaf area index and reflectance in the middle infrared bands. The figures show growth stage accounting for most of the difference between the regression functions. The inconsistencies between locations of the slopes and y-intercepts of the regression functions were probably due to background noise in the sensor, and differences in sunlight intensity and in sun angle.
The amount of variability in total N uptake as explained by NDVI increased with advancing growth stages in the long-term N & P fertility trial at Lahoma and the variety by N rate trial at Hennessey (Figures 2 and 3). In the variety by N rate trial, total N uptake expressed higher correlation with spectral radiance at growth stages 5 and 7 than at growth stage 4 (Figure 3). As growth stage increased, so too did the percentage of soil covered by vegetation. In the variety by N rate trial, in January, the percent of soil cover was less than 50%, leading to a high degree of soil reflectance interference in the spectral radiance readings. Thus, it is thought that the optical sensing systems ability to detect N deficiencies was impaired when soil cover was less than 50%. As percent cover increased, background soil reflectance should have decreased. Thus, NIR reflectance increased and red reflectance decreased (Knipling, 1970; Gausman et al., 1976; and Walburg et al., 1982).
In the long term N & P fertility trial at Stillwater, a higher correlation with NDVI existed for Feekes growth stage 5 than for Feekes growth stage 7. In addition to growth stage, environmental factors impacted spectral radiance readings. From January 18th through the 24th, air temperatures were below 0°C causing cell blasting. The resulting grayish foliage in the variety by N rate trial may have increased the Jan 25th red spectral radiance. This further explains the low correlation between NDVI and total N uptake observed at the variety by N rate trial at Feekes growth stage 4. Secondly, drought injury was significant for the 1995 Oklahoma winter wheat crop. Plants under drought stress can accumulate NO3 proline, a free amino acid that may act as a storage vessel for nitrogen (Tyankova, 1967) and ammonium. In addition, initial soil nitrate and ammonium levels were high in the variety by N rate trial. Subsequently, the variety by N rate data from Feekes growth stage 7 showed forage N concentrations as high as 5%.
The freeze in the variety by N rate trial just prior to the Jan. 25th sensor reading and harvest many have led to the same amount of biomass present at both Feekes growth stage 4 (January) and 5 (February). Perhaps the freeze contributed to no increase in biomass. In January, even though the cells had lost turgor, much of the vegetative material was still there. By February, the leaves had either recovered or died. The amount of dead vegetation led to the low biomass present at Feekes growth stage 5. New vegetative growth had not surpassed the amount that had died. By April, the crop had accumulated biomass above its level in January and February.
Furthermore, correlation between grain N concentration and NDVI existed at Feekes growth stage 7, but not at Feekes growth stages 4 and 5. At Feekes growth stages 4 and 5, inorganic N concentration was 2000 to 3000 mg kg-1, but declines to 200 to 300 mg kg-1 by Feekes growth stage 7. Further research was done to ascertain this observation.
Previous research has shown forage NO3N to be higher at earlier stages of growth stage and declining with advancing stage of growth (Roth et al., 1989). As NO3N is converted to amino acids with advancing stages of growth, NO3N concentrations should decrease. Analysis using cadium reduction followed by automated flow injection of the forage samples showed that at Feekes growth stage 4 samples averaged 350 mg kg-1. NO3N concentrations averaged 1200 mg kg-1 for Feekes growth stage 5 and 1192 mg kg-1 for Feekes growth stage 7, which was not consistent with previous literature.
Total forage N concentrations for Feekes growth stages 4, 5, and 7 as analyzed by Carlo Erba (Milan, Italy) NA 1500 dry combustion analyzer, averaged 4.5, 4.7 and 4.8 %, respectively. The ratio of nitrate to total N was expected to decrease with advancing growth stage as organic N levels increase. Deepening of chlorophyll color and an increase in the number of vibrational bonds may have increased NDVI values. The increase in biomass also was expected to decrease background interference. Therefore, we expected to find improved correlation of NDVI and forage N concentration with advancing stage of growth due to increased relative amounts of organic N.
ConclusionS
Spectral readings of winter wheat were affected by its growth stage. At lower growth stages, NDVI could not predict total N uptake in the forage. Correlation increased as percent soil cover increased. N rate had a linear effect on NDVI and total N uptake. Freeze injury impaired the ability of NDVI to predict total N uptake. Since the slope and y-intercept of the function describing the relationship between NDVI and total N uptake changed with respect to time and location, the calibration curve used to apply topdress N fertilizer must be generated on a per growth stage and per field basis. Future research is necessary to discover at which growth stage VRT based N fertilizer should be applied to obtain the highest economic return on grain and forage yields.
References
Gausman, H.W., W.A. Allen, D.E. Escobar, R.R. Rodriguez, and R. Cardenas. 1971. Age effects of cotton leaves on light reflectance, transmittance, and absorptance and on water content and thickness. Agron. J. 63:465-469.
Hagger, R.J., C.J. Stent, and J. Rose. 1984. Measuring spectral differences in vegetation canopies by a reflectance ratio meter. Weed Research, 24:59-65.
Hinzman, L.D., M.E. Bauer, C.S.T. Daughtry. 1986. Effects of nitrogen fertilization on growth and reflectance characteristics of winter wheat. Remote Sens. Envir., 19:47-61.
Jackson, R.D., P.N. Slater, P.J. Pinter, Jr. 1983. Discrimination of growth and water stress in wheat by various vegetation indices through clear and turbid atmospheres. Remote Sens. of Envir., 13:187-208.
Kleman, J. and E. Fagerlund. 1987. Influence of different nitrogen and irrigation treatments on the spectral reflectance of barley. Remote Sens. Envir. 21:1-14.
Knipling, E.B. 1970. Physical and physiological basis for the reflectance of visible and near-infrared radiation from vegetation. Remote Sens. of Envir. 1:155-159.
Large, E.C. 1954. Growth stages in cereals. Plant Pathol. 3:128-129.
Roth, G.W., R.H. Fox, and H.G. Marshall. 1989. Plant tissue tests for predicting nitrogen fertilizer requirements of winter wheat. Agron. J. 81:502-507.
Sawyer, J.E. 1994. Concepts of variable rate technology with considerations for fertilizer application. J. Prod. Agric. 7:195-201.
Solie, J.B., W.R. Raun, R.W. Whitney, M.L. Stone, and J.D. Ringer. 1996. Optical sensor based field element size and sensing strategy for nitrogen application. In press, Transactions of ASAE.
Stone, M.L., J.B. Solie, W.R. Raun, R.W. Whitney, S.L. Taylor and J.D. Ringer. 1996. Use of spectral radiance for correcting in-season fertilizer nitrogen deficiencies in winter wheat. Trans. ASAE 39(5):1623-1631.
Tyankova, L.A. 1967. Distribution of the free and bound proline and of the free hydroxyproline in the separate organs of wheat plants during drought. C.R. Acad. Bulg. Sci. 20:583-586.
Walburg, G., M.E. Bauer, C.S.T. Daughtry, and T.L. Housley. 1982. Effects of nitrogen nutrition on growth, yield, and reflectance characteristics of corn canopies. Agron. J. 74:677-683.
Walter-Shea, E.A., J.M. Norman, B.L. Blad, and B.F. Robinson. 1991. Leaf reflectance and transmittance in soybean and corn. Agron. J. 83:631-636.
Wanjura, D.F. and J.L. Hatfield. 1987. Sensitivity of spectral vegetative indices to crop biomass. Transactions of the ASAE 30(3):810-815.
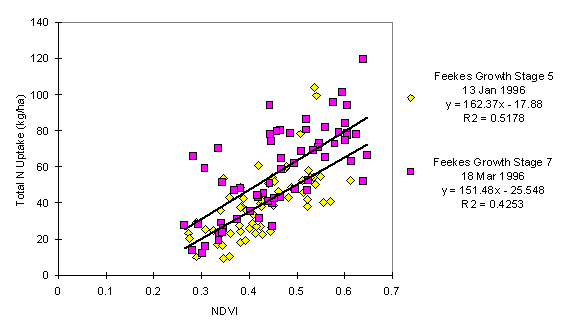
Figure 1. Regression of Total N Uptake in Winter Wheat on the Normalized Difference Vegetative Index (NDVI) at two Feekes Growth Stages (5 and 7), Stillwater, OK, 1996.
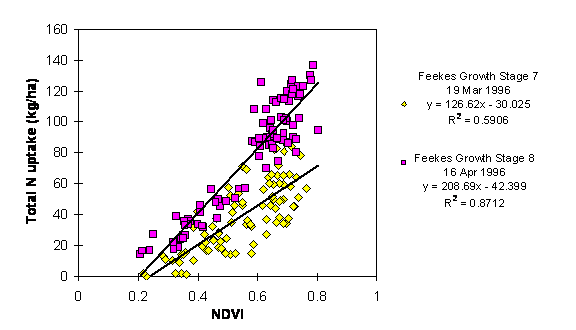
Figure 2. Regression of Total N Uptake in Winter
Wheat on the Normalized Difference Vegetative Index (NDVI) at two
Feekes Growth Stages (7 and 8),
Lahoma, OK, 1996.

Figure 3. Regression of Total N Uptake in Winter Wheat on the Normalized Difference Vegetative Index (NDVI) at three Feekes Growth Stages (4, 5, and 7), Hennessey, OK, 1996.
Table 1. Initial surface (0 - 15cm) soil test characteristics at each location.
__________________________________________________________________________
pH NH4-N NO3-N P K OC TN
-------------------------mg kg-1----------------------- ------------g kg-1---------
Hennessey 5.8 18.3 21.1 142 674 12.3 1.00
Lahoma 5.0 45 320 7.1 0.62
Stillwater 5.8 70 211 6.4 0.67
__________________________________________________________________________
pH, 1:1 soil:deionized water; K and P, Mehlich-3.
Table 2. Analysis of variance for forage N uptake, NDVI, red, green, and NIR at Stillwater, OK.
_____________________________________________________________________________
Sources of variation df N uptake (kg ha-1) NDVI Red Green NIR
_____________________________________________________________________________
Mean squares
Replication 3 263 0.012 327497** 686710** 1748064*
N rate 3 1475* 0.044 75981 29939 345628
Growth stage 1 2270** 0.008* 554447* 2104222* 5568846*
N rate*growth stage 3 152 0.003 35659 72445 298302
N rate linear 1 4294** 0.122* 169033@ 1015 808857
N rate quadratic 1 106 0.007 56160 84185 102702
Means
Nitrogen rate, kg ha-1
0 36.56 0.397 777.92 1114.43 1751.64
45 41.71 0.402 813.29 1200.56 1895.20
90 54.42 0.489 723.40 1237.83 2205.32
134 66.86 0.552 591.20 1118.80 2122.27
SED 7.3 0.057 157.75 258.2 452.72
Growth Stage
5 41.46 0.444 594.82 911.47 1576.44
7 58.31 0.476 858.09 1424.33 2410.77
_____________________________________________________________________________
@,*, **, *** Significant at the 0.10, 0.05, 0.01, and 0.001 probability levels, respectively.
SED = standard error of the difference between two equally replicated treatment means.
Table 3. Analysis of variance for forage N uptake, NDVI, red, green, and NIR at Lahoma, OK
_____________________________________________________________________________
Sources of variation df N uptake (kg ha-1) NDVI Red Green NIR
_____________________________________________________________________________
Mean squares
Replication 3 774* 0.011 51358 161328 794218
N rate 5 4746*** 0.149*** 1675323*** 1005994* 647491
Growth stage 1 9690*** 0.001 767404** 3481413** 13272296***
N rate*growth stage 5 552** 0.002 64712 186044 847411
N rate linear 1 22189*** 0.710*** 7354401*** 3971062** 1495830
N rate quadratic 1 494 0.029 654624 352564 110531
Means
Nitrogen rate, kg ha-1 GS7 GS8
0 16.38 24.38 0.355 1637.89 2228.10 3398.61
22 19.93 36.71 0.432 1401.79 2097.86 3501.94
45 39.23 56.8 0.549 813.19 1498.15 2798.36
67 52.61 88.35 0.637 690.53 1580.68 3078.61
90 44.3 94.05 0.657 661.66 1565.05 3126.71
112 58.17 100.83 0.701 502.12 1352.35 2844.14
SED 5.46 5.46 0.053 151.05 265.11 (NS)
_____________________________________________________________________________
*, **, *** Significant at the 0.05, 0.01, and 0.001 probability levels, respectively, NS Not significant.
SED = standard error of the difference between two equally replicated treatment means. GS7 and GS8, Feekes growth stages 7 and 8, respectively.
Table 4. Analysis of variance for forage total N uptake, NDVI, red, green, and NIR, Hennessey, OK.
_____________________________________________________________________________
Sources of variation df N uptake NDVI Red Green NIR
_____________________________________________________________________________
Mean squares
Replication 2 2932.02** 0.0632** 82664.24* 2870.69 184059
N rate 2 710.69 0.0251 88372.87* 31742.06 3608
Variety 4 1801.46** 0.0144 94998.66** 19690.57 97936
N rate*variety 8 255.48 0.005 62433.97* 60263.00*** 131896
Rep*variety*N rate(a) 28 432.47 0.0111 22996.93 12485.70 82530
Growth stage 2 152700.64** 2.16** 1966798** 10030973** 70568654**
N rate*growth stage 4 363.24 0.0103 26256.56 8993.2 8297
Variety*growth stage 8 735.37** 0.0099 47915.17 46718.1 163431*
N rate*variety*growth stage 16 432.14 0.0059 26324.04 42962.5 148560*
_____________________________________________________________________________
*, **, *** Significant at the 0.05, and 0.01probability levels, respectively.
Table 5. Effect of variety and growth stage on forage total N uptake, NDVI, red, green, and NIR, Hennessey, OK.
_____________________________________________________________________________
Variety Growth N uptake NDVI Red Green NIR
Stage kg/ha
_____________________________________________________________________________
Treatment means
Agri-Pro "Hickok" 4 15.6 0.1468 1000.0 1146.9 1348.2
5 22.3 0.3627 639.1 886.6 1393.1
7 116.7 0.5268 1079.1 1862.9 3499.3
AGSECO 7853 4 16 0.1289 967.7 1092.3 1258.9
5 26.8 0.3746 642.8 914.4 1403.3
7 140.8 0.6672 771.9 1829.3 3829.9
Jagger 4 42.8 0.1785 973.6 1149.7 1403.1
5 22.5 0.407 555.6 815 1307.5
7 158 0.64 825.2 1763.1 3753.3
Karl 92 4 23.2 0.1653 1006.5 1180.2 1412.9
5 23.9 0.3615 467.2 663.5 998.9
7 138 0.6593 766.1 1863.2 3661.6
Tonkawa 4 18.6 0.1405 1002.4 1145.3 1332.9
5 29.1 0.4003 604.1 872.5 1382.9
7 123.3 0.6112 915.5 1841.3 3754.1
SED 9.8 0.0362 76.5 97.7 181.7
_____________________________________________________________________________
SED = standard error of the difference between two equally replicated treatment means.